ALM Ortho, Inc.
Advanced 3D Printed Orthopedic Implants with fastest Time to Mark...
Private Fundraise
This company may be interested in raising funds from accredited investors. You must Request Access to see more information about this company.
Request Access 9 Company Overview
Company Overview

ALM Ortho is a technology company specializing in developing 3D printed titanium orthopedic (implants) devices. In partnering with and with a plan to acquire Additive Manufacturing supplier, ALM Ortho can now deliver the highest-quality, customized implant solutions in industry-leading Time-to-Market (TTM) of 2-weeks.
Key Highlights:
- 2 spine devices being prepared for FDA clearance and will be commercialization ready Q1 2021
- Acquire additive-manufacturing market leader Amplify Additive
- Intellectual property acquired for Spine and Long Bone fixation
- Partnered with the University of Texas, El Paso for new material for Ortho
- ALM owns and controls all IP related to new products
 Time-to-Market Delays
Time-to-Market Delays

Orthopedic devices are always associated with long time-to-market durations.
Device Customization Ideal
Orthopedic implant solutions required device customization for ideal patient outcomes. Often the customization process includes an extensive TTM delay period.
Infrastructure Required

 Disorganized Infrastructure
Disorganized Infrastructure
Previously, the technology or the ability to assemble all these processes efficiently under one roof has left both patients and practitioners waiting on long TTM durations.
The end-product often takes countless months on average.
 Fastest Ever TTM
Fastest Ever TTM

ALM Ortho delivers customized orthopedic devices to market in record times.

 Customized Designed Infrastructure
Customized Designed Infrastructure
We use additive manufacturing to develop customized implant devices efficiently at scale.
 Our TTM
Our TTM
We deliver the most advanced, reliable, implant devices to market in 2 weeks or less.
 Full Design and Manufacturing Oversight
Full Design and Manufacturing Oversight
We will acquire Amplify Additive, the leading additive manufacturing supply company, resulting in unmatched advantages in the marketplace.
 Keeping Patients In-mind
Keeping Patients In-mind
Through process oversight, we deliver patients comfort at speed they deserve.
 Our Customs Process
Our Customs Process

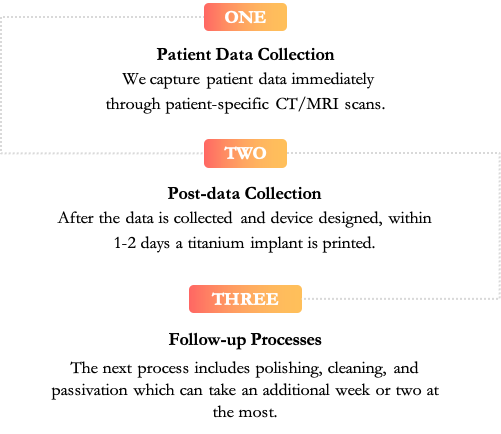
 Our Mfg Process
Our Mfg Process

 Additive Manufacturing Explained
Additive Manufacturing Explained
Employing advanced designs with lattice structures enabling advanced fusion.
Powder Bed Fusion Technology
A powder bed fusion technology called Electron Beam Melting(EBM) melts fine layers of titanium powder. The process allows orthopedic implant devices to be customized for each patient.
 Partnerships
Partnerships


 Intellectual Property
Intellectual Property

 Spine and Long Bone Fixation
Spine and Long Bone Fixation
(Trauma and Oncology)
- Two FDA approved spine devices are being readied for commercialization
- Additional IP for Spine and Long Bone fixation has been acquired
- MVP has been completed
- Mechanical submission work pertaining to FDA submission is being completed.

Additional Intellectual Property
In-development
- SI Joint implant system
- Total Hip stem
- New material for Total Knee implant.
 Meet the Team
Meet the Team


Co-founder
- Recognized as a leading US KOL for Orthopedic implant design & development leveraging AM
- 2014 helped to introduced the Arcam Q10Plus platform to US Orthopedic market
- Managed largest AM OEM Ortho. Supplier group, DiSanto/GE Additive
- Founder of 2nd largest Ortho AM supplier, Amplify Additive
- Original founder of Additive Orthopedics, Leading Implant Company for 3D printed Foot & Ankle implants
- Presented to the FDA on AM & contributed to FDA Guidance Document on Additive Manufacturing
- Assisted with first FDA 510(K) cleared Cranial plate on Arcam Q10 Platform
- Designed & Developed five FDA 510(K) cleared implants for Foot & Ankle indications
Co-founder
- Seasoned senior executive with significant experience in guiding companies through periods of growth and transition. A diverse background leading both small and large companies, public and private, in a variety of industries.
- Specialties: sales and marketing, corporate finance, strategic planning, mergers and acquisition execution and integration, going public, financial and operating controls, team building
Co-founder
- Investor & VP Business Development Amplify
- Extensive clinical experience in orthopedic OR setting
- Experience with FDA requirements for 3D Printed implants
- Experienced sales consultant for 3D printed custom & standard orthopedic implants on Arcam platform
- ●Experience in Oncology and Veterinary implant indications

 Team Members Joining Soon...
Team Members Joining Soon...

VP of Personalized Medical Devices
- Extensive clinical experience in orthopedic OR settings providing technical support to physicians
- Extensive experience creating custom solutions for the shoulder, hip, knee, ankle, elbow, wrist, long bones and spine.
- Over 4,000 patient-specific Custom orthopedic devices
- Extensive medical device experience with R&D, FDA and EU regulations pertaining to custom devices, and Quality System regulations & design controls.
VP, National Sales Director, Orthopedics
- Performance driven sales executive, creating demand for medical devices, manage, hire, train, motivate, and direct the selling efforts of extremity sales teams in the United States. Manage direct reports. Annually exceed growth goals. Oversee all sales functions including sales force management, distribution channels, sales programs, forecasting and sales force integration.
VP, Application Engineering for AM
- Recognized as a Leading Application Engineer for the 3D Printing technology & for orthopedics in US & Europe
- 2014 helped to introduced the Arcam Q10Plus platform to US Orthopedic market
- Managed largest AM OEM Ortho. Supplier group, Oerlikon (formally, GE Additive/DiSanto Technology)

The Business Plan area is locked. You must Request Access to this company to see more information including the Business Plan.
Request Access
N
No updates yet.
%
No backers yet.
Quick Signup TBD
You must have an account to do this!
